Clinical Pipeline
A single, secure platform to design, run, and release clinical CART batches with full electronic traceability and regulatorygrade documentation.
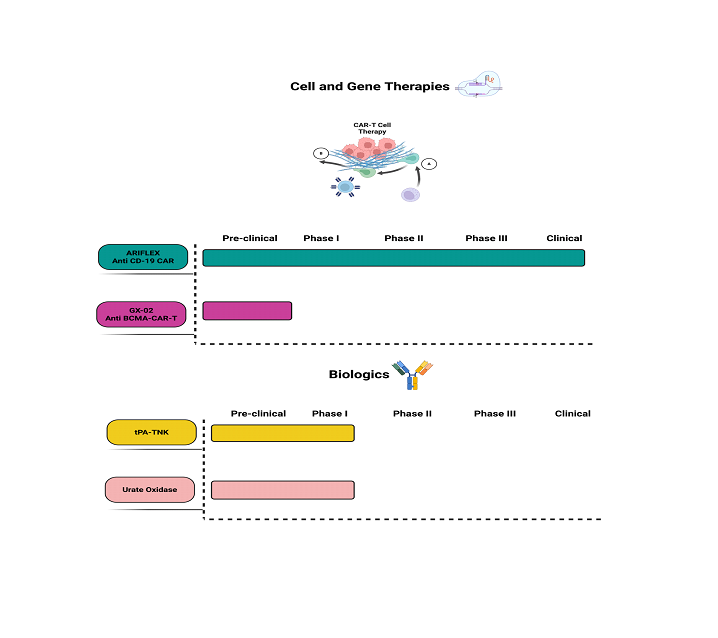
Cell and Gene Therapy
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease
Our LogicGate platform enables precise cellular programming for superior therapeutic delivery and performance
Supported by a modular manufacturing system, we provide scalable and economical production of next generation cell and gene therapy products.
Biologics
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease.
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease.
Software for Clinical Cell and Gene Therapy Production:
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease.
Clinical Grade Reagents (GMP)
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease.
The Cambridge GenetiX CART Cell Extensive Characterization Panel is a multicolor flow cytometry panel designed to profile CART phenotype, differentiation, and activation status in a single assay. It supports inprocess monitoring during manufacturing, lotrelease characterization, and longitudinal patientsample analysis, providing a highcontent view of product quality.
A typical panel configuration can include:
⦁ Lineage and panT markers: CD3, CD4, CD8, CD45 (Tcell identification and major subsets).
⦁ Memory and differentiation markers: CD45RA, CD45RO, CCR7, CD62L, CD27, CD28, CD95(naïve, central memory, effector memory, TEMRA, and stemlike memory definition).
⦁ Activation and costimulation markers: CD69, CD25, 41BB (CD137), OX40 (CD134), HLADR(early and sustained activation, costimulatory engagement).
⦁ Proliferation/fitness markers: Ki67 (cycling status), optional inclusion of CD127 (IL7Rα) for survival potential.
⦁ CAR tracking and Bcell exclusion: antiCAR detection reagent (compatible with your CAR construct) and CD19 or other target antigen where relevant.
Used together, these markers allow you to resolve CART products into functionally meaningful subsets (e.g., CD4 vs CD8, naïve vs memory, activated vs resting) and to track how the phenotype evolves across the manufacturing process or after infusion. This panel is intended for research use and can be adapted to your preferred fluorochrome backbone and cytometer configuration
At Cambridge GenetiX, we engineer therapeutic cells designed to transform and potentially cure human disease.
CAR-T Potency Test kits
CGx TruTarget CAR Antigen Bead Set – Antigen‑coated particles that provide stable, lot‑to‑lot‑consistent surrogates for CAR‑T potency and assay control.
